诚信认证:
工商注册信息已核实!
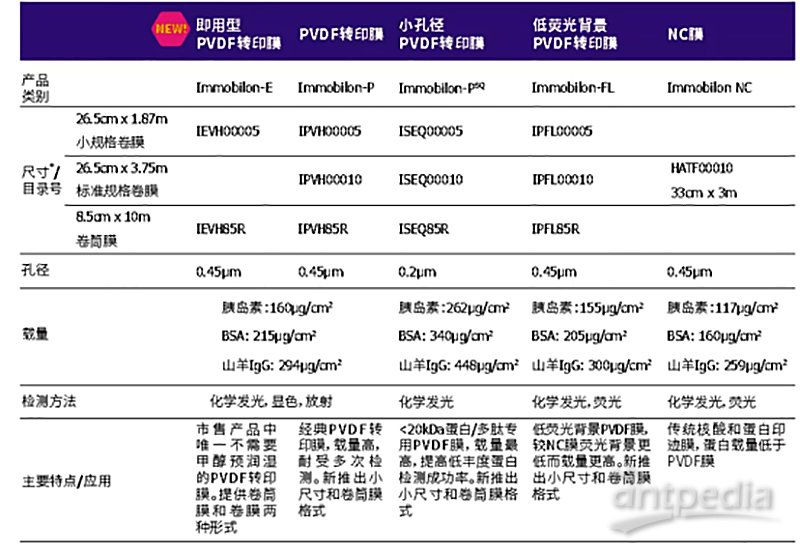
ISEQ00010 Immobilon-PSQ 小孔径PVDF转印膜
| 品牌 | 厂商性质 | 产地 | 货期 |
|---|---|---|---|
| Millipore | 生产商 | 德国 | 订货 |
性能特点
货号:ISEQ00010
| 货号 | 规格 | 操作 |
|---|---|---|
| ISEQ00010 | 1 EA | 询底价 |
ISEQ00010 Millipore
Immobilon-PSQ PVDF 膜
1 roll, 26.5 cm x 3.75 m, 0.2 µm pore size, Hydrophobic PVDF Transfer Membrane for western blotting.
eCl@ss: 32031602
物料
PVDF membrane
plain filter
white filter
质量水平
400
特点
hydrophobic
manufacturer/tradename
Immobilon®
technique(s)
western blot: suitable
过滤器长度 × 宽度
26.5 cm × 3.75 m
孔径
0.2 μm pore size
容量
262 μg/cm2 adsorption capacity (insulin)
340 μg/cm2 adsorption capacity (BSA)
448 μg/cm2 adsorption capacity (goat IgG)
相容性
for use with Amido black
for use with CPTS
for use with Colloidal gold
for use with Coomassie brilliant blue
for use with India ink
for use with Ponceau-S red
for use with Sypro<TMSYMBOL></TMSYMBOL> ruby
for use with Toluidine blue
for use with Transillumination
detection method
chemiluminescent
colorimetric
radioactive
运输
ambient
说明
一般描述
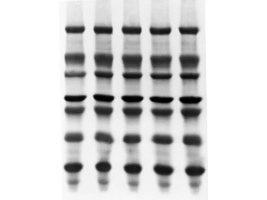
The Immobilon®-PSQ membrane is a 0.2 μm microporous membrane made up of polyvinylidene fluoride (PVDF). It has been developed to maximize protein binding. Immobilon®-PSQ membrane is recommended for direct protein sequencing and immunodetection following electroblotting of protein from electrophoresis gels (or dot binding of purified protein samples), especially for proteins with molecular weights less than 20,000. This membrane is compatible with tank and semi-dry electroblotting systems.
应用
Immobilon®-PSQ PVDF Membrane has been used in western blot.
特点和优势
Immobilon®-PSQ Membrane:
0.2 μm pore size and a large internal structure
Higher protein adsorption and sequencing yields than other membranes
Prevents blow-through of low molecular weight proteins
法律信息
Immobilon is a registered trademark of Merck KGaA, Darmstadt, Germany
安全信息
储存分类代码
11 - Combustible Solids
WGK
WGK 3
闪点(F)
Not applicable
闪点(C)
Not applicable
ISEQ00010 Immobilon-PSQ 小孔径PVDF转印膜信息由默克生命科学为您提供,如您想了解更多关于ISEQ00010 Immobilon-PSQ 小孔径PVDF转印膜报价、型号、参数等信息,欢迎来电或留言咨询。
注:该产品未在中华人民共和国食品药品监督管理部门申请医疗器械注册和备案,不可用于临床诊断或治疗等相关用途